Advanced low-temperature solid oxide fuel cells based on a built-in electric field
Solid oxide fuel cells (SOFCs) show considerable promise for meeting the current ever-increasing energy demand and environmental sustainability requirements as a result of their high efficiency and low environmental impact. To enable high ionic conductivity, SOFCs are often required to operate at high temperature, which in turn results in high costs[1]. Therefore, lowering the operational temperatures has become a major priority in SOFC research and development[2]. According to the traditional concepts of SOFCs, single semiconductor materials are usually considered as electrolyte membrane due to their higher ionic conductivity, with heterostructures constructed from different semiconductor materials having never been considered. Recently, Meng et al.[3] made an important breakthrough in low-temperature SOFCs by introducing semiconductor heterojunction membranes to function alternatively as electrolytes with better performance. This novel fuel cell design is known as a semiconductor-ionic membrane fuel cell (SIMFC)[3-5]. Zhang et al.[6], Nie et al.[7], Deng et al.[8], Mushtaq et al.[9], and Afzal[10] used semiconductor materials, including Ni0.8Co0.15Al0.05LiO2-δ[6], La0.6Sr0.4Co0.2Fe0.8O3-δ[7], Sr2Fe1.5Mo0.5O6-δ[8], SrFe0.75Ti0.25O3-δ[9],
Recently, Wang et al.[18] reported a 3C-SiC, which was tuned for protonic conducting properties via the construction of an n-p heterostructure composite with Na0.6CoO2, exhibiting an ionic conductivity of
It is well known that semiconductor materials have already been successful in photovoltaic cells based on a built-in electric field (BIEF)[23]. Generally, when p- and n-type semiconductors are contacted, the redistribution of charges at the interface constitutes a space-charge region with the BIEF pointing from the n- to p-type region[24]. The BIEF is also applied in lithium-ion batteries. Qiao et al.[25] demonstrated a BIEF to reduce the space charge layer formation and boost lithium-ion transport in all-solid-state lithium-ion batteries by an in-situ differential phase contrast scanning transmission electron microscopy technique and finite element method simulations.
Most importantly, Zhang et al.[6], Nie et al.[7], Deng et al.[8], Mushtaq et al.[9], and Afzal[10] successfully applied semiconductor materials in SOFCs by compositing semiconductor and ionic conductor materials to construct SIMFCs with BIEF effects. To understand the principle of SIMFCs, a physical mode based on a BIEF and alignment of the energy band, similar to a perovskite solar cell (PSC), was proposed by
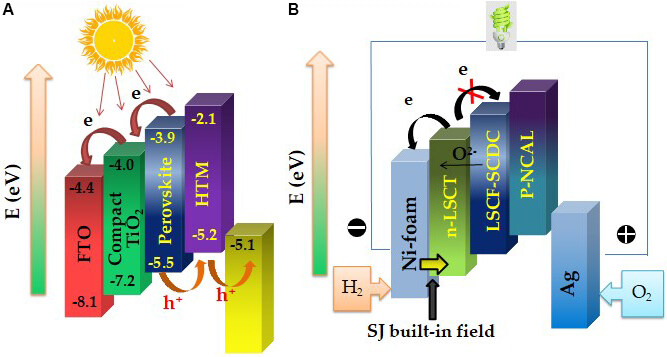
Figure 1. Energy level diagrams of (A) a PSC and (B) the fuel-to-electricity conversion device inspired by the PSC structure[16]. LSCT:
Interestingly, a Schottky junction (SJ) was also found in this all new device, where Ni0.8Co0.15Al0.05LiO2-δ (NCAL) is reduced into a Ni-Co alloy at the anode with H2, as indicated in Figure 1B. After the SJ is formed between the anode and electrolyte, it can also inhibit electrons passing through the electrode and SM while simultaneously enhancing the transportation of H+ or O2- due to the BIEF formation at the interface. The used materials for PSC and SIMFC devices are listed in Table 1. It is true that the junction plays a vital role in blocking electrons crossing over the internal device to avoid short circuiting and also in promoting the ionic transport process.
Comparison of materials used for PSCs and SIMFCs
| Back contact | Electron transport layer | Functional layer | Hole transport layer | Metal electrode | |
| PSC | Fluorine-doped tin dioxide (FTO) | TiO2 | Perovskite absorber | Hole-transporter materials | Au |
| SIMFC | Ni foam | La0.2Sr0.25Ca0.45TiO3 | La0.6Sr0.4Co0.2Fe0.8O3-δ-Sm and Ca co-doped CeO2 | Ni0.8Co0.15Al0.05LiO2-δ | Ag |
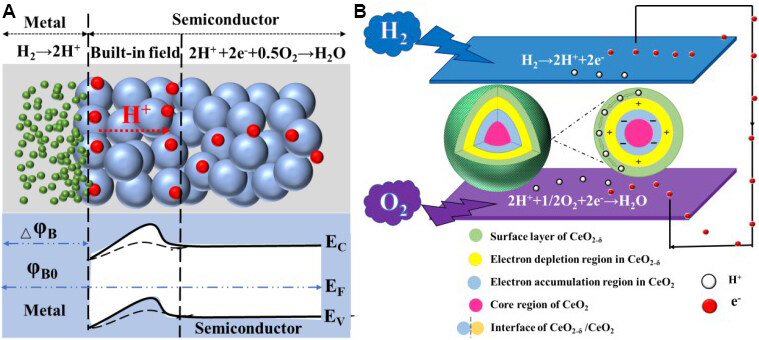
According to our previous research, the BIEF can be formed by a Schottky heterojunction[24], intrinsic-negative (i-n) heterojunction[12] and p-n heterojunction[25]. In a Schottky heterojunction, the BIEF can be built up simply at the interface of the metal (electrode) and semiconductor (electrolyte) regions. Yun et al.[28] constructed SJ fuel cells using a p-type semiconductor material, namely, a LiNi0.85Co0.15O2-δ (LCN) composite with Ce0.8Sm0.2O1.9-Na2CO3, which exhibited a high power output of 1000 mW cm-2 at 550 °C. A thin Ni-metal layer originating from reduction of the semiconducting oxide LCN is formed at the H2 side. Therefore, a Ni-metal/p-type LCN-semiconductor SJ is formed that can accelerate ion transport capacity while inhibiting electrons from passing through the junction formed at the anode/semiconductor membrane interface, as shown in Figure 2A.
In order to prove the as-reported SJ, Zhu et al.[29] tested the response current as a function of bias voltage for a half cell with the structure of NCAL-Ni/7LCP-3ZnO at 550 °C with air and a H2 flow to the NCAL electrode side. The results indicated that an apparent rectification response emerged in the I-V characteristic analogous to the reported I-V response of a Ni/ZnO-polar contact, illustrating a Schottky contact between the reduced anode and membrane layer. Furthermore, the characterization of semiconductor properties and band structures is very important for SIMFCs based on semiconductor-ionic membranes with BIEF effects, which are very different from conventional SOFCs based on ionic conducting electrolytes. Therefore, various new characterization techniques from semiconductor aspects, like ultraviolet-visible spectroscopy[30], ultraviolet photoelectron spectroscopy[30], Hall coefficient measurements[30], density functional theory calculations[31] and so on, have been introduced to determine the band structures and prove the BIEF effects on SIMFCs. The different characteristics of SIMFCs compared to SOFCs are obvious because of the use semiconductor-based membranes vs. conventional electrolytes.
In an i-n heterojunction, an i-n type interface contact is constructed, e.g., a CeO2/CeO2-δ core-shell heterostructure, where CeO2 is an intrinsic i-type semiconductor and CeO2-δ is an n-type semiconductor. A charge separation occurs at the CeO2-δ/CeO2 interface. The electrons can transfer from the shell to the core while it is forbidden to pass through from the core to the shell. After the CeO2/CeO2-δ core-shell heterostructure forms, an electron depletion region is built on the CeO2-δ side at the interface and an electron accumulation region is simultaneously formed on the CeO2 side of the interface. On this basis, the local charge distribution and the electric field or the BIEF are formed among particle surfaces, which stop protons from migrating deep inside the shell to pass through the interface and suppress the bulk infiltration of the surface protons, as shown in Figure 2B. Benefiting from the BIEF, a “proton shuttle” is constructed in the continuous highly conducting regions formed in the ceria-semiconductor membrane of the SOFC.
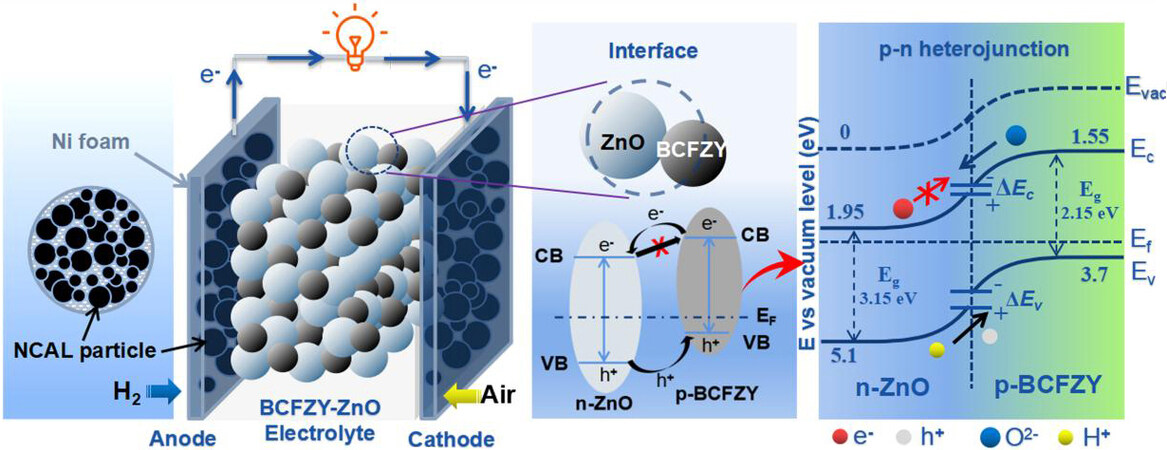
To further understand the new mechanism in SIMFCs, Xia et al.[30] constructed a BaCo0.4Fe0.4Zr0.1Y0.1O3-δ (BCFZY)-ZnO p-n heterostructure for low-temperature SOFCs. They found that the as-prepared heterostructure exhibits a hybrid H+/O2- conducting capability during fuel cell operation. When two semiconductors with different energy band levels are combined, conduction band offset (ΔEc) and valence band offset (ΔEv) will be induced, thus forming potential barriers to form the BIEF. To explain its mechanism, a BIEF in the as-prepared BCFZY-ZnO p-n heterostructure was introduced, where the junction prevented the electron passing through and the ionic conductivity can be enhanced by the BIEF, as illustrated by Figure 3.
Figure 3. Schematic diagram of a typical p-n heterojunction formed at the heterophasic interface of a p-type (BCFZY)-n-type (ZnO) semiconductor membrane and the corresponding energy band alignment mechanism proposed for interpreting the charge separation and ionic transportation process[30]. BCFZY: BaCo0.4Fe0.4Zr0.1Y0.1O3-δ; NCAL: Ni0.8Co0.15Al0.05LiO2-δ; CB: conduction band; VB: valence band.
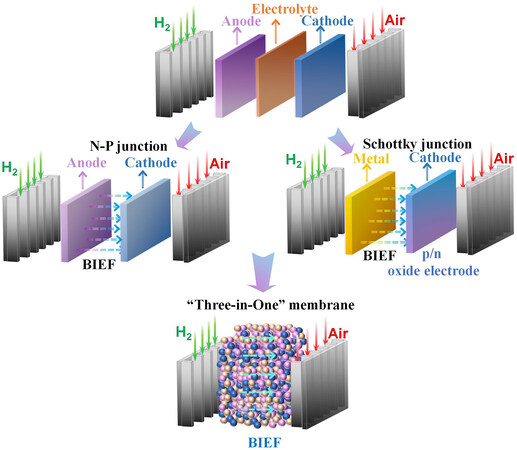
Cai et al.[32] explained the SIMFC device from the conventional three layers of anode/electrolyte/cathode to a “three in one” membrane, as shown in Figure 4. A semiconductor-ionic membrane based on NiO-yttrium-stabilized zirconia (YSZ)-LSCF composites was used to construct a SIMFC device. Under H2/air conditions, the SJ BIEF is formed that can avoid the electronic short-circuit problem. As a result, A high power density of 680 mW·cm-2 at 600 °C was achieved with an open circuit voltage of 1.11 V.
Figure 4. From conventional three layered fuel cells to a “three in one” membrane with a BIEF. BIEF: Built-in electric field.
Encouraged by this new concept, Cai et al.[33] further constructed a bulk heterostructure nanocomposite electrolyte of Ce0.8Sm0.2O2-δ-SrTiO3 for low-temperature SOFCs, exhibiting a peak power density of
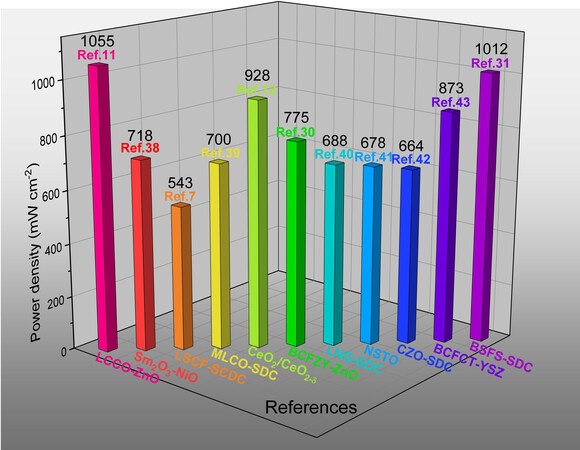
Overall, significant progress has been achieved in the field of SIMFCs due to the innovation of semiconductor materials as membranes and advances in forming the BIEF. A high power density of
Figure 5. Summary of previous work on SIMFCs. LCCO-ZnO: La/Pr co-doped CeO2-ZnO; LSCF-SCDC: La0.6Sr0.4Co0.2Fe0.8O3-δ-Sm and Ca co-doped CeO2; MLCO-SDC: Mg-doped LiCoO2-Sm doped CeO2; CeO2/CeO2-δ: the core hall structure of the CeO2/CeO2-δ; BCFZY-ZnO: BaCo0.4Fe0.4Zr0.1Y0.1O3-δ-ZnO; LNO-SDC: LaNiO3-Sm doped CeO2; NSTO: Nb-doped SrTiO3-δ; CZO-SDC: Co0.2Zn0.8O-Sm-doped CeO2; BCFCT-YSZ: BaCo0.2Fe0.1Ce0.2Tm0.1-Zr0.3Y0.1O3-δ; BSFS-SDC: BaSrFeSbO3-Sm doped CeO2.
As shown in Table 1, a Voc of > 1.0 V was individually realized in different heterostructures, suggesting that the performance of devices can be fully improved by the BIEF produced from semiconductor heterostructures. To achieve this goal, the development of high-performance SIMFCs is of significant importance. It is encouraging that the long-term stability of SIMFCs can reach over 100 h, according to recent reports [Table 2]. We believe that the as-reported SIMFCs will provide a new route for SOFC research and development towards commercialization. Compared with traditional SOFCs, SIMFCs exhibit various advantages, e.g., high ionic conductivity resulting in higher performances, including current and power outputs at low temperature, simple structures (three in one) and so on. However, in future research and development, some critical perspectives are suggested:
Long-term stability data for SIMFCs from recent reports
| Semiconductor membrane | VOC (V) | Stability (h) | Temp. (°C) | Year | Ref. |
| La/Pr co-doped CeO2-ZnO | 1.04 | - | 550 | 2018 | [11] |
| Sm2O3-NiO | 1.04 | 2 | 550 | 2018 | [38] |
| La0.6Sr0.4Co0.2Fe0.8O3-δ-Sm and Ca co-doped CeO2 | 1.05 | - | 550 | 2019 | [7] |
| Mg-doped LiCoO2-Sm doped CeO2 | 1.02 | 10 | 600 | 2019 | [39] |
| CeO2/CeO2-δ core-shell structure | 1.04 | - | 520 | 2019 | [12] |
| BaCo0.4Fe0.4Zr0.1Y0.1O3-δ-ZnO | 1.01 | ~2 | 550 | 2020 | [30] |
| LaNiO3-Sm doped CeO2 | 1.02 | 52 | 530 | 2020 | [40] |
| Nb-doped SrTiO3-δ | 1.03 | - | 520 | 2021 | [41] |
| Co0.2Zn0.8O-Sm-doped CeO2 | 1.06 | 35 | 550 | 2021 | [42] |
| BaCo0.2Fe0.1Ce0.2Tm0.1-Zr0.3Y0.1O3-δ | 1.07 | 100 | 530 | 2021 | [43] |
| BaSrFeSbO3-Sm doped CeO2 | 1.09 | 100 | 550 | 2021 | [31] |
(1) Long-term stability is currently absent from engineering efforts with regards to commercialization. More effort should be contributed to the engineering and scaling-up of SIMFCs;
(2) In order to develop long-term SIMFC durability, the development of compatible electrode materials has made good progress;
(3) Relevant theoretical models and calculations should be employed to guide further research and development. In particular, the physical properties and effect of the BEIF formed from various heterojunctions, e.g., bulk and planar p-n, Schottky, n-i (intrinsic or insulating) junctions;
(4) Some new technologies and technical processes combined with SOFCs and protonic ceramic fuel cells should be introduced to develop durable SIMFCs.
The first demonstration of SOFC technology was made in the 1930s by Baur and Preis[36] and used zirconia stabilized with 15 wt.% of yttria (the so-called Nernst Mas) as the electrolyte, iron or carbon as the anode and magnetite (Fe3O4) as the cathode. Long durability could not be achieved until the compatible electrodes, NiO-YSZ cermet and especially perovskite oxide cathode materials, were discovered and technically developed to incorporate with the YSZ electrolyte. This took several decades. Compatible electrodes for semiconductor-ionic material membranes have yet to be employed into SIMFCs. Nevertheless, significant progress has been made in this area.
It is also noteworthy that SIMFCs are built not only on electrochemistry but also semiconductor physics[37]. We expect that this report can arouse significant attention from related research fields and disciplines to overcome the bottleneck of SOFC commercialization.
DECLARATIONS
AcknowledgmentsThis work was supported by Southeast University (SEU) Solar Energy and Joint Energy Storage Center, Functional Materials Laboratory (FML), Xi’an University of Architecture and Technology (XAUAT), and Laboratory of Functional materials and device, Nanjing Xiaozhuang University.
Authors’ contributionsMade substantial contributions to conception and design of this Research Highlight: Yun S, Zhu B
Investigation, formal analysis, writing - original draft: Lu Y
Data curation: Shi J
Supervision, methodology, resources, visualization, funding acquisition, project administration, writing - review & editing: Yun S, Zhu B
Availability of data and materialsNot applicable.
Financial support and sponsorshipThis work was supported by NSFC (No. 51672208, 51772080), Key Program for International S&T Cooperation Projects of Shaanxi Province (No. 2019KWZ-03, No. 2019JZ-20), and Open foundation Project of key Laboratory of Plateau Green Building and Ecological Community of Qinghai Province (No. KLKF-2019-002) is greatly acknowledged. This research was also funded by the Foundation of Nanjing Xiaozhuang University (Grant No. 2020NXY12).
Conflicts of interestAll authors declared that there are no conflicts of interest.
Ethical approval and consent to participateNot applicable.
Consent for publicationNot applicable.
Copyright© The Author(s) 2021.
REFERENCES
1. Fan L, Zhu B, Su P, He C. Nanomaterials and technologies for low temperature solid oxide fuel cells: recent advances, challenges and opportunities. Nano Energy 2018;45:148-76.
2. Zhang Y, Knibbe R, Sunarso J, et al. Recent progress on advanced materials for solid-oxide fuel cells operating below 500 °C. Adv Mater 2017;29:1700132.
3. Meng Y, Mi Y, Xu F, et al. Low-temperature fuel cells using a composite of redox-stable perovskite oxide La0.7Sr0.3Cr0.5Fe0.5O3-δ and ionic conductor. J Power Sources 2017;366:259-64.
4. Li J, Lu Y, Li D, Qi F, Yu L, Xia C. Effects of P-N and N-N heterostructures and band alignment on the performance of low-temperature solid oxide fuel cells. Int J Hydrogen Energy 2021;46:9790-8.
5. Wang B, Wang Y, Fan L, et al. Preparation and characterization of Sm and Ca co-doped ceria-La0.6Sr0.4Co0.2Fe0.8O3-δ semiconductor-ionic composites for electrolyte-layer-free fuel cells. J Mater Chem A 2016;4:15426-36.
6. Zhang W, Cai Y, Wang B, et al. The fuel cells studies from ionic electrolyte Ce0.8Sm0.05Ca0.15O2-δ to the mixture layers with semiconductor Ni0.8Co0.15Al0.05LiO2-δ. Int J Hydrogen Energy 2016;41:18761-8.
7. Nie X, Chen Y, Mushtaq N, et al. The sintering temperature effect on electrochemical properties of Ce0.8Sm0.05Ca0.15O2-δ (SCDC)-La0.6Sr0.4Co0.2Fe0.8O3-δ (LSCF) heterostructure pellet. Nanoscale Res Lett 2019;14:162.
8. Deng H, Feng C, Zhang W, et al. The electrolyte-layer free fuel cell using a semiconductor-ionic Sr2Fe1.5Mo0.5O6-δ - Ce0.8Sm0.2O2-δ composite functional membrane. Int J Hydrogen Energy 2017;42:25001-7.
9. Mushtaq N, Xia C, Dong W, et al. Tuning the energy band structure at interfaces of the SrFe0.75Ti0.25O3-δ-Sm0.25Ce0.75O2-δ heterostructure for fast ionic transport. ACS Appl Mater Interfaces 2019;11:38737-45.
10. Afzal M, Saleemi M, Wang B, et al. Fabrication of novel electrolyte-layer free fuel cell with semi-ionic conductor (Ba0.5Sr0.5Co0.8Fe0.2O3-δ-Sm0.2Ce0.8O1.9) and Schottky barrier. J Power Sources 2016;328:136-42.
11. Shah M, Mushtaq N, Rauf S, Xia C, Zhu B. The semiconductor SrFe0.2Ti0.8O3-δ-ZnO hetero-structure electrolyte fuel cells. Int J Hydrog Energy 2019;44:30319-27.
12. Xing Y, Wu Y, Li L, et al. Proton shuttles in CeO2/CeO2-δ core-shell structure. ACS Energy Lett 2019;4:2601-7.
13. Chen G, Liu H, He Y, et al. Electrochemical mechanisms of an advanced low-temperature fuel cell with a SrTiO3 electrolyte. J Mater Chem A 2019;7:9638-45.
14. Islam QA, Paydar S, Akbar N, Zhu B, Wu Y. Nanoparticle exsolution in perovskite oxide and its sustainable electrochemical energy systems. J Power Sources 2021;492:229626.
15. Zhu B, Lund P, Raza R, et al. A new energy conversion technology based on nano-redox and nano-device processes. Nano Energy 2013;2:1179-85.
16. Zhu B, Huang Y, Fan L, et al. Novel fuel cell with nanocomposite functional layer designed by perovskite solar cell principle. Nano Energy 2016;19:156-64.
17. Zhu B, Raza R, Liu Q, et al. A new energy conversion technology joining electrochemical and physical principles. RSC Adv 2012;2:5066.
18. Wang F, Xing Y, Hu E, et al. PN heterostructure interface-facilitated proton conduction in 3C-SiC/Na0.6CoO2 electrolyte for fuel cell application. ACS Appl Energy Mater 2021;4:7519-25.
19. Lu Y, Mi Y, Li J, Qi F, Yan S, Dong W. Recent progress in semiconductor-ionic conductor nanomaterial as a membrane for low-temperature solid oxide fuel cells. Nanomaterials (Basel) 2021;11:2290.
20. Xu D, Yan A, Xu S, et al. Self-Assembled Triple (H+/O2-/e-) conducting nanocomposite of Ba-Co-Ce-Y-O into an electrolyte for semiconductor ionic fuel cells. Nanomaterials (Basel) 2021;11:2365.
21. Zhu B, Mi Y, Xia C, et al. Nano-scale view into solid oxide fuel cell and semiconductor membrane fuel cell: material and technology. Energy Mater 2021;1:2.
22. Fang Y, Dong Q, Shao Y, Yuan Y, Huang J. Highly narrowband perovskite single-crystal photodetectors enabled by surface-charge recombination. Nature Photon 2015;9:679-86.
23. Assadi M, Bakhoda S, Saidur R, Hanaei H. Recent progress in perovskite solar cells. Renew Sustain Energy Rev 2018;81:2812-22.
24. Choi S, Kucharczyk CJ, Liang Y, et al. Exceptional power density and stability at intermediate temperatures in protonic ceramic fuel cells. Nat Energy 2018;3:202-10.
25. Qiao Z, Xia C, Cai Y, et al. Electrochemical and electrical properties of doped CeO2-ZnO composite for low-temperature solid oxide fuel cell applications. J Power Sources 2018;392:33-40.
26. Wang L, Xie R, Chen B, et al. In-situ visualization of the space-charge-layer effect on interfacial lithium-ion transport in all-solid-state batteries. Nat Commun 2020;11:5889.
27. Yun S, Qin Y, Uhl AR, et al. New-generation integrated devices based on dye-sensitized and perovskite solar cells. Energy Environ Sci 2018;11:476-526.
28. Yun S, Zhou X, Even J, Hagfeldt A. Theoretical treatment of CH3NH3PbI3 perovskite solar cells. Angew Chem Int Ed Engl 2017;56:15806-17.
29. Zhu B, Lund PD, Raza R, et al. Schottky junction effect on high performance fuel cells based on nanocomposite materials. Adv Energy Mater 2015;5:1401895.
30. Xia C, Mi Y, Wang B, Lin B, Chen G, Zhu B. Shaping triple-conducting semiconductor BaCo0.4Fe0.4Zr0.1Y0.1O3-δ into an electrolyte for low-temperature solid oxide fuel cells. Nat Commun 2019;10:1707.
31. Mushtaq N, Lu YZ, Xia C, et al. Promoted electrocatalytic activity and ionic transport simultaneously in dual functional Ba0.5Sr0.5Fe0.8Sb0.2O3-δ-Sm0.2Ce0.8O2-δ heterostructure. Applied Catalysis B: Environmental 2021;298:120503.
32. Cai Y, Wang B, Wang Y, et al. Validating the technological feasibility of yttria-stabilized zirconia-based semiconducting-ionic composite in intermediate-temperature solid oxide fuel cells. J Power Sources 2018;384:318-27.
33. Cai Y, Chen Y, Akbar M, et al. A bulk-heterostructure nanocomposite electrolyte of Ce0.8Sm0.2O2-δ-SrTiO3 for low-temperature solid oxide fuel cells. Nanomicro Lett 2021;13:46.
34. Zhang Y, Liu J, Singh M, et al. Superionic conductivity in ceria-based heterostructure composites for low-temperature solid oxide fuel cells. Nanomicro Lett 2020;12:178.
35. Hu E, Jiang Z, Fan L, et al. Junction and energy band on novel semiconductor-based fuel cells. iScience 2021;24:102191.
37. Zhu B, Fan L, Mushtaq N, et al. Semiconductor electrochemistry for clean energy conversion and storage. Electrochemical Energy Reviews 2021; doi: 10.1007/0-306-48036-0_4.
38. Liu L, Liu Y, Li L, Wu Y, Singh M, Zhu B. The composite electrolyte with an insulation Sm2O3 and semiconductor NiO for advanced fuel cells. Int J Hydrogen energy Energy 2018;43:12739-47.
39. Ganesh KS, Wang B, Kim J, Zhu B. Ionic conducting properties and fuel cell performance developed by band structures. J Phys Chem C 2019;123:8569-77.
40. Lu Y, Akbar M, Xia C, et al. Catalytic membrane with high ion-electron conduction made of strongly correlated perovskite LaNiO3 and Ce0.8Sm0.2O2-δ for fuel cells. Journal of Catalysis 2020;386:117-25.
41. Shah MAKY, Rauf S, Zhu B, et al. Semiconductor Nb-doped SrTiO3−δ perovskite electrolyte for a ceramic fuel cell. ACS Appl Energy Mater 2021;4:365-75.
42. Rauf S, Shah MAKY, Zhu B, et al. Electrochemical properties of a dual-ion semiconductor-ionic Co0.2Zn0.8O-Sm0.20Ce0.80O2−δ composite for a high-performance low-temperature solid oxide fuel cell. ACS Appl Energy Mater 2021;4:194-207.
Cite This Article
Export citation file: BibTeX | RIS
OAE Style
Lu Y, Zhu B, Shi J, Yun S. Advanced low-temperature solid oxide fuel cells based on a built-in electric field. Energy Mater 2021;1:100007. http://dx.doi.org/10.20517/energymater.2021.06
AMA Style
Lu Y, Zhu B, Shi J, Yun S. Advanced low-temperature solid oxide fuel cells based on a built-in electric field. Energy Materials. 2021; 1(1): 100007. http://dx.doi.org/10.20517/energymater.2021.06
Chicago/Turabian Style
Lu, Yuzheng, Bin Zhu, Jing Shi, Sining Yun. 2021. "Advanced low-temperature solid oxide fuel cells based on a built-in electric field" Energy Materials. 1, no.1: 100007. http://dx.doi.org/10.20517/energymater.2021.06
ACS Style
Lu, Y.; Zhu B.; Shi J.; Yun S. Advanced low-temperature solid oxide fuel cells based on a built-in electric field. Energy Mater. 2021, 1, 100007. http://dx.doi.org/10.20517/energymater.2021.06
About This Article
Copyright
Data & Comments
Data
 Cite This Article 26 clicks
Cite This Article 26 clicks













Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.