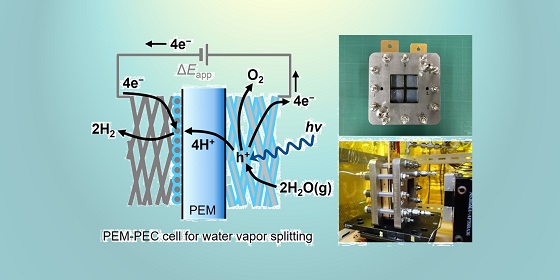
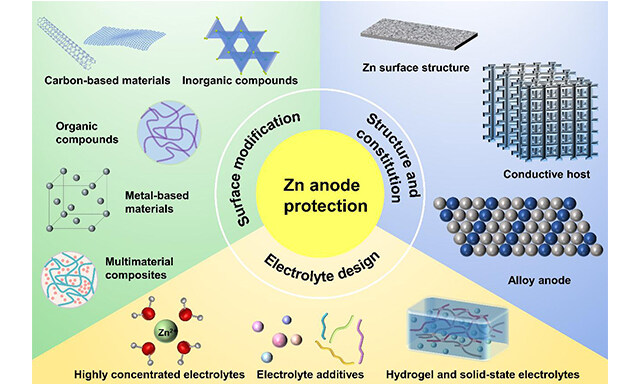
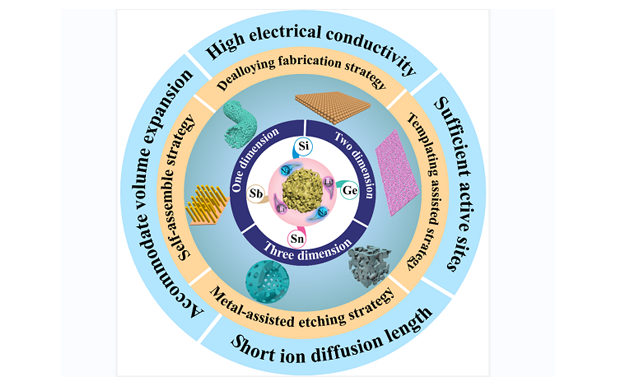
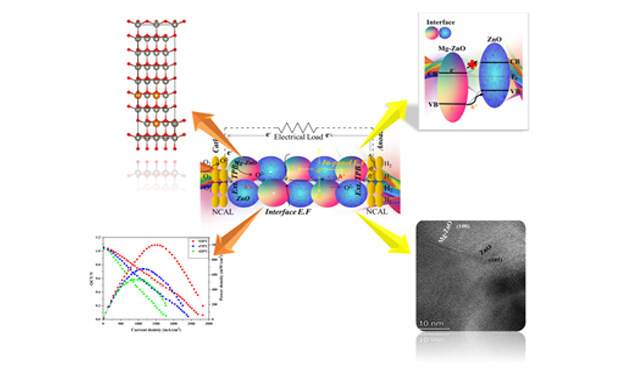
Energy Materials
Views: Downloads:
Views: Downloads:
Views: Downloads:
Views: Downloads:
Views: Downloads:
Views: Downloads:
Views: Downloads:
Views: Downloads:
Data
1002
Authors
633
Reviewers
2021
Published Since
1,885,165
Article Views
307,385
Article Downloads
For Reviewers
For Readers
Add your e-mail address to receive forthcoming Issues of this journal:
Themed Collections
Volume 4, Issue 3 (in progress)
Volume 4, Issue 2
Volume 4, Issue 1
Volume 3, Issue 6
Volume 3, Issue 5
Volume 3, Issue 4
Volume 3, Issue 3
Volume 3, Issue 2
Volume 3, Issue 1
Volume 2, Issue 6
Volume 2, Issue 5
Volume 2, Issue 4
Volume 2, Issue 3
Volume 2, Issue 2
Volume 2, Issue 1
Volume 1, Issue 2
Volume 1, Issue 1
Related Journals
Related Journals
Data
1002
Authors
633
Reviewers
2021
Published Since
1,885,165
Article Views
307,385
Article Downloads